Published
Stability-Indicating RP-HPLC Method Development for A Fixed-Dose Combination Tablet and Its Degradation Kinetics
Published in JPRIMS, Vol-2, Issue-8, Aug.-2025 (Vol. 2, Issue 8, 2025)
Abstract
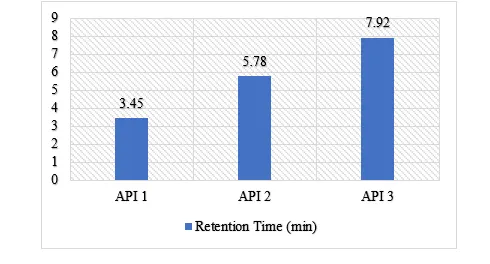
Pharmaceutical formulations stability is an important factor in drug development and drug quality control. The goals of this work were to establish and qualify a stability-indicating reverse-phase high-performance liquid chromatography (RP-HPLC) method to measure active pharmaceutical ingredients in a fixed- dose combination admixture tablet simultaneously and to assess the degradation kinetics of the Active pharmaceutical ingredients under different stress conditions. The concentration using C18 column and acetonitrile-phosphate buffer mobile phase proved to be optimal in terms of separating the APIs with great resolution, symmetric peaks and reproducibility. The results of the forced degradation studies indicated that stress due to acidic and oxidative conditions appeared to be the most significant stressors, whereas thermal and photolytic stress had little effect. First-order kinetic models were used to describe the degradation that was confirmed to be significantly different under different stress conditions using statistical testing. The validated technique was robust and sensitive and could be applied in regular quality checks; the information helped understand the stability of the formulation and shelf-life.
Authors (1)
Sanjeev Kumar
ESIC Medical College and Hospi...ESIC Medical College and Hospital, Bihta, Patna, B...ESIC Medical College and Hospital, Bihta, Patna, Bihar, Pin No - 80110...ESIC Medical College and Hospital, Bihta, Patna, Bihar, Pin No - 801103
View all publications →Download Article
Best for printing and citation
File size: 0.0 MB
Format: PDF
Download Article
Best for printing and citation
File size: 0.0 MB
Format: PDF
Article Information
JPRIMS820149
JPRIMS-01-000149
Article Impact
Views:5,792
Downloads:2,333
How to Cite
, S. (2025). Stability-Indicating RP-HPLC Method Development for A Fixed-Dose Combination Tablet and Its Degradation Kinetics. Journal of Pharmaceutical Research and Integrated Medical Sciences, 2(8), xx-xx. https://jprims.scholarjms.com/articles/JPRIMS820149
Article Actions
More from this Issue
Formulation And Evaluation of Herbal Inhalation Pods to Relief Nasal Congestion
Gurtej Singh Wadhwa, Suma...Read more →
IMPACT OF HYPERTENSION AND DIABETES ON THE PATIENTS OF LOCAL AREAS OF DURG CITY
Tarun Kumar Sahu, Saurab...Read more →
