Formulation and Evaluation of Multilayer Matrix Tablet of Captopril forOral Controlled Delivery
Published in April 2025 (Vol. 2, Issue 4, 2025)
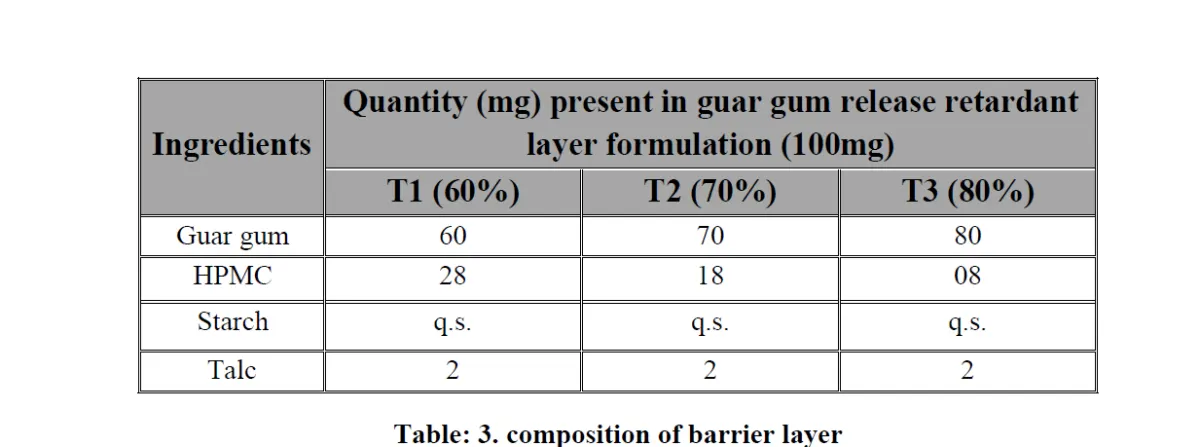
Abstract
Authors (3)
Deepak Biswas
KIPS, Shrishankaracharya Profe...KIPS, Shrishankaracharya Professional University, ...KIPS, Shrishankaracharya Professional University, (C.G). India KIPS, Shrishankaracharya Professional University, (C.G). India
View all publications →Vamini Madhukar
Shri Rawatpura Sarkar Institut...Shri Rawatpura Sarkar Institute of Pharmaceutical ...Shri Rawatpura Sarkar Institute of Pharmaceutical Sciences, Kumhari, C...Shri Rawatpura Sarkar Institute of Pharmaceutical Sciences, Kumhari, Chhattisgarh, India
View all publications →Arpan Trpathi
FPS, SSTC, Bhilai, Chhattisgar...FPS, SSTC, Bhilai, Chhattisgarh, India FPS, SSTC, Bhilai, Chhattisgarh, India FPS, SSTC, Bhilai, Chhattisgarh, India
View all publications →Download Article
Best for printing and citation
Download Article
Best for printing and citation
Article Information
Published in:
April 2025 (Vol. 2, Issue 4, 2025)JPRIMS420058
JPRIMS-01-000058
Article Impact
How to Cite
, D. & , V. & Trpathi (2025). Formulation and Evaluation of Multilayer Matrix Tablet of Captopril forOral Controlled Delivery. Journal of Pharmaceutical Research and Integrated Medical Sciences, 2(4), xx-xx. https://jprims.scholarjms.com/articles/JPRIMS420058
Article Actions
More from this Issue
Nicotine Dependence Diagnosis Methods
Applications of Quantum Chemistry in Molecular Spectroscopy and Reactivity
Green Synthesis And Characterization OfMeteal Nanopartical For CatalyticApplication
More by These Authors
Niosome-Based Approaches in Ocular Drug Delivery: Advances, Challenges, And Future Perspectives
• Vol. , Issue
Read more →